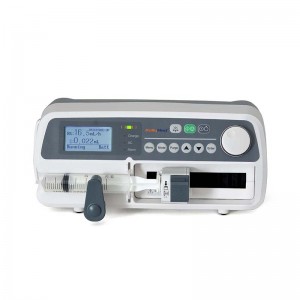
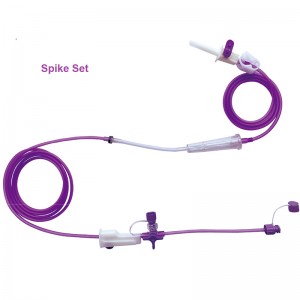
product
Focus on medical equipment for 27 years
about us
About factory description

what we do
Founded in 1994, Beijing KellyMed Co., Ltd. is a high technology corporation engaged in R&D, manufacturing and marketing of infusion systems, supported by Institute of Mechanics, Chinese Academy of Sciences. Manufacturing Facility, R&D Center, QC Division, Domestic Sales Division, Int’l Sales Division and Customer Support Center were established under KellyMed. Engineers are major in Physics, Infrared Radiation, Electronics, Ultrasound, Automation, Computer, Sensor and Mechanics.
Our newsletters, the latest information about our products, news and special offers.
Click for manual-

technology
New technology transformation mode,research high quality products
-

Research
Professional research project team for different customer needs
-

personnel
The company introduces a large number of talents, researches projects and is responsible for customers

application
Lorem ipsum dolor sit amet, consectetur adipisicing elit
-

ICU
-

NICU
-
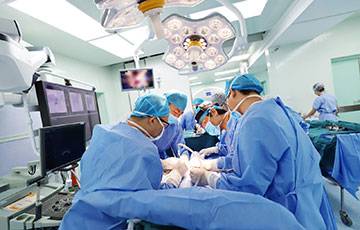
Operation Room
-

Nursing Home
-
 30
30
Years In Medical
-
 400+
400+
Kellymed Staff
-
 60+
60+
Countries
-
 50000
50000
Installations
-
 100+
100+
Overseas Distributors
news
Lorem ipsum dolor sit amet, consectetur adipisicing elit